Immunohistochemistry Protocol Frozen Section
Fix sections in dry acetone for 15 minutes.
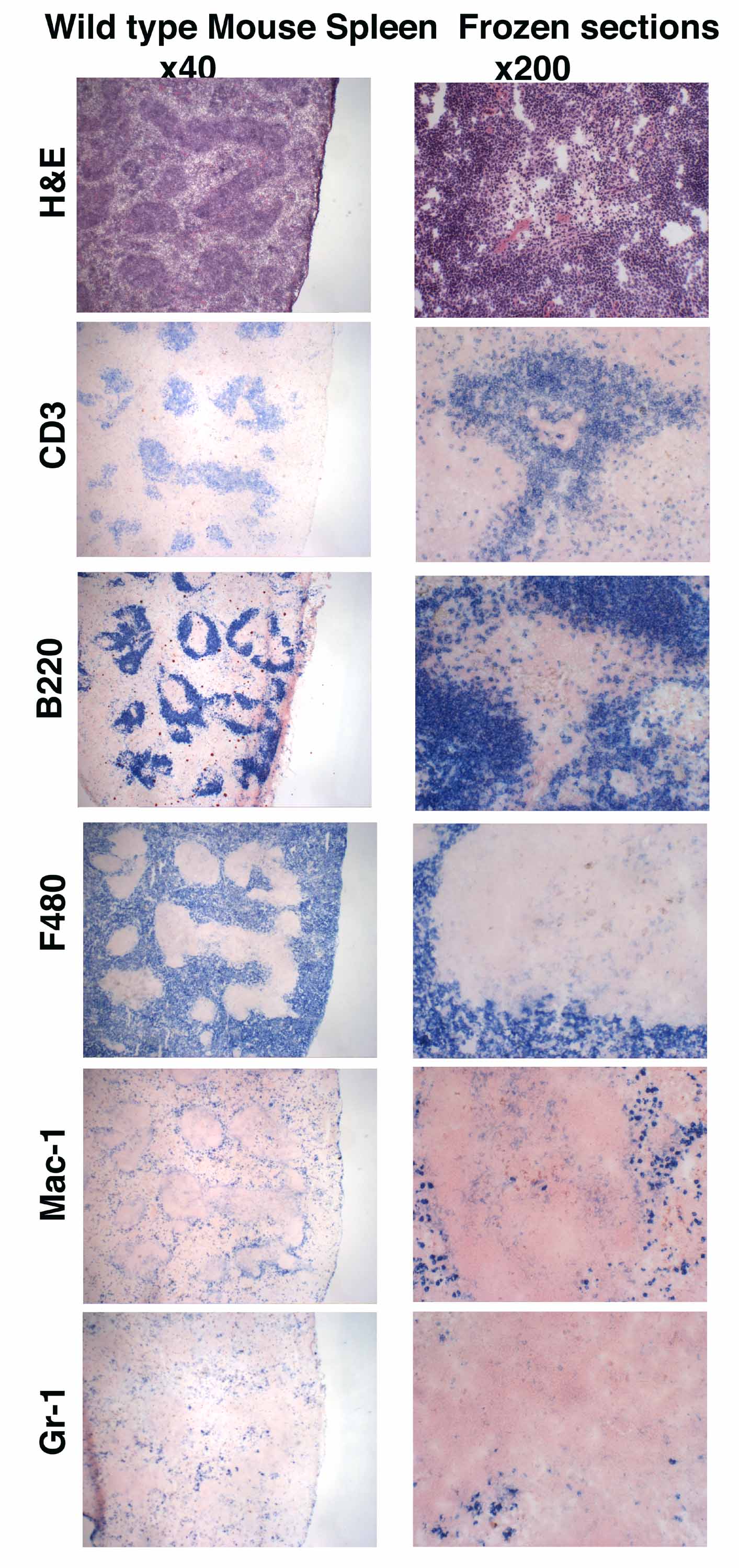
Immunohistochemistry protocol frozen section. Place the tissue in a small sealed box if the tissue is too small to hold it pour in some embedding reagent like oct. Section tissue at a range of 6 8 µm and place on positively charged slides. This may prevent cracking of the block when sectioning.
In most cases bio rad recommends that tissues are snap frozen in liquid nitrogen then prepared as 4 6 μm sections using a cryostat. For tissue stored at 80 c. Each investigator must determine the precise experimental conditions required to generate a strong and specific signal for each antigen of interest.
Allow to evaporate for 10 minutes. This ihc protocol provides a basic guide for the fixation cryostat sectioning and staining of frozen tissue samples. Apply primary antibody diluted in tbs with 1 bsa.
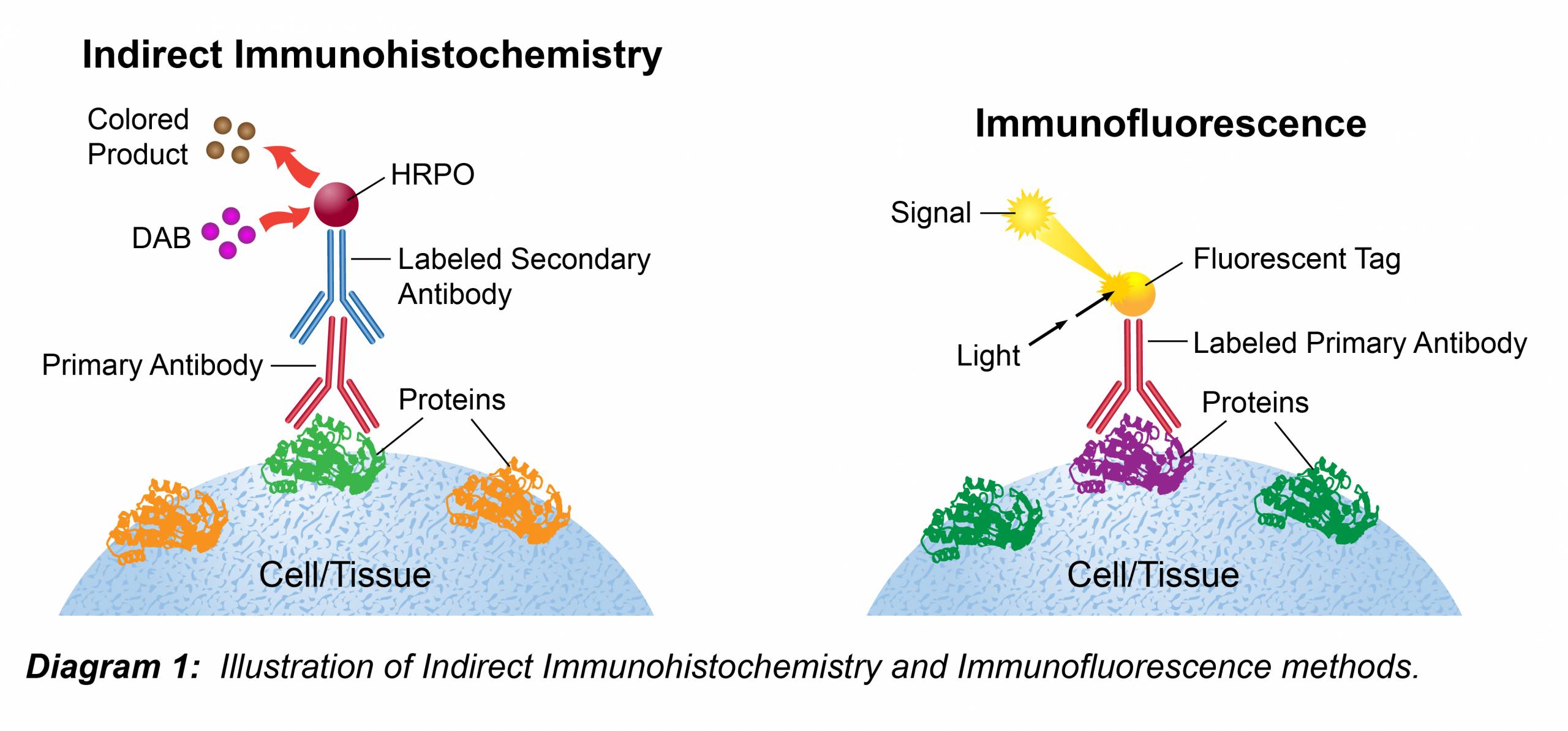
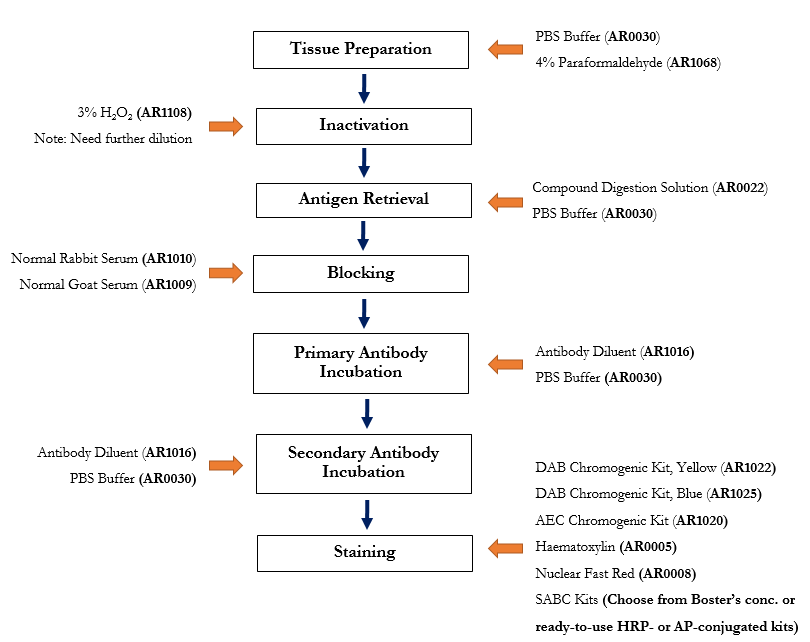
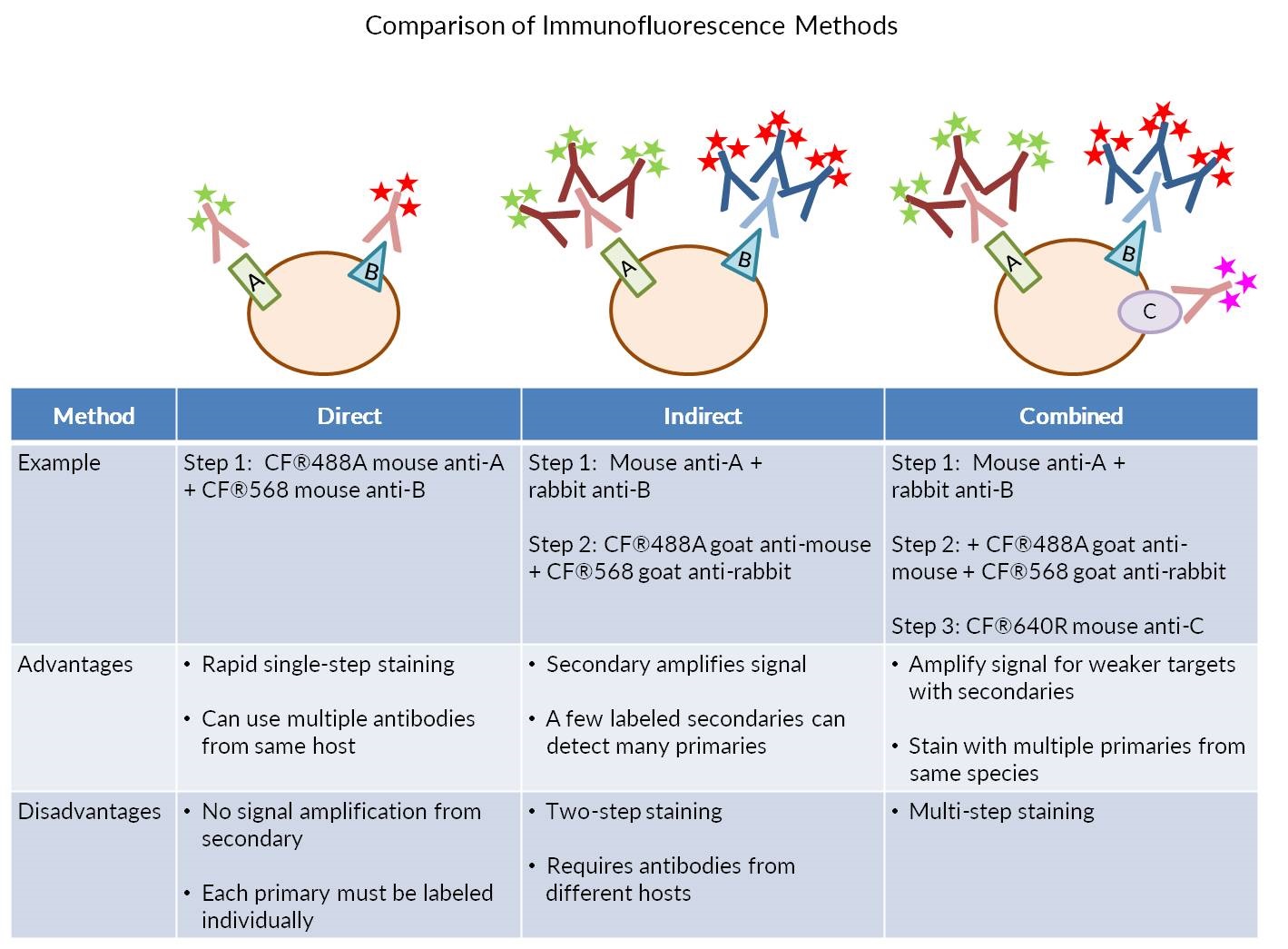
The following immunohistochemistry ihc protocol has been developed and optimized by r d systems ihc icc laboratory for chromogenic ihc experiments using frozen tissue samples. Transfer the tissue to frozen section machine or store at 80 c for future use. The fluorescent immunohistochemistry immunofluorescence protocol below is intended for the fluorescent visualization of protein expression in frozen tissue sections.
Remove from freezer and equilibrate at 20 c for approximately 15 minutes before attempting to section. Sections can be stored in a sealed slide box at 80 c for later use. In my experience when the frozen section is sitting cold on the stage the effect of drying is minimal.
Drain slides for a few seconds do not rinse and wipe around the sections with tissue paper. This ihc protocol provides a basic guide for the fixation cryostat sectioning and staining of frozen tissue samples. The following immunohistochemistry ihc protocol has been developed and optimized by r d systems ihc icc laboratory for fluorescent ihc experiments using frozen tissue samples.