H E Staining Protocol For Frozen Sections

Optimal staining is achieved with 5 6 µm thick sections.
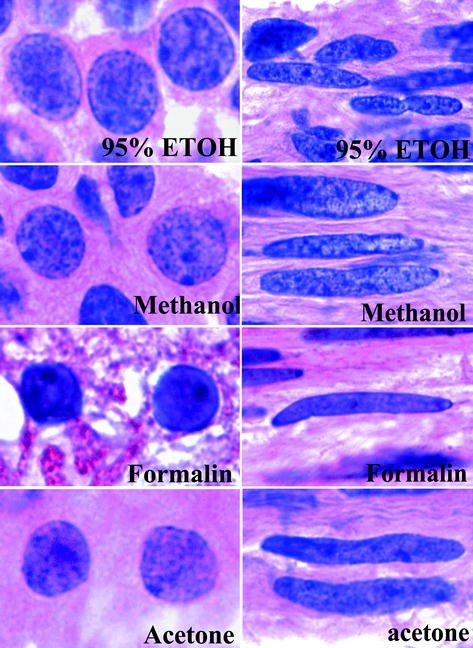
H e staining protocol for frozen sections. Transfer to formalin 10 phosphate buffered 1090 for 10 dips. After obtaining frozen section immediately fix in 95 ethanol even 15 seconds of delay can cause significant artifact formal alcohol formalin or 95 alcohol. If hamatoxylin is stored in 50 ml tube it should be wrapped in foil.
H e haematoxylin and eosin staining for frozen tissue sections. Pour off the fixative and allow acetone to evaporate from the tissue sections for 20 min at room temperature. Rinse well in distilled water.
5 7 seconds hematoxylin. Change staining solutions on a regular basis according to acceptable laboratory protocol. Stain with filtered 0 1 mayers hematoxylin sigma.
The format is easily reproduced and the reagents resilient enough to allow for large numbers of slides to be stained consistently before reagents need to be changed. Do not allow frozen sections to air dry at any time during the staining procedure for better preservation of tissue morphology. Hard tap water or 1 ammonium hydroxide in order to neutralize the acid and form an insoluble blue aluminium haematin complex.
Immediately fix frozen sections in 95 ethyl alcohol for 15 seconds. H e haematoxylin and eosin staining for frozen tissue sections. Air dry sections for several minutes to remove moisture.
15 20 seconds. During staining alum haematoxylin stained sections are usually passed on to a neutral or alkaline solution e g. See procedure note 1.